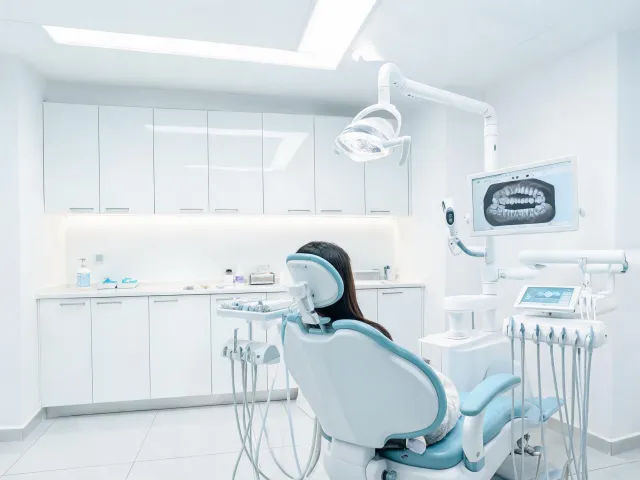
Dental Groups
Imaging, sterilization, chairside diagnostics, and documentation packs for multi-site dental operations.
Dental, diagnostic, and imaging systems for practical clinical teams
From chairside dental workflows to central lab automation and radiology suites, Danaher organizes device selection around the people who install, validate, clean, connect, and use equipment every day.
Care settings
Procurement teams can start with the clinical setting, then narrow choices by service coverage, interoperability, documentation package, and training burden.

Imaging, sterilization, chairside diagnostics, and documentation packs for multi-site dental operations.

IVD analyzers, middleware planning, QC routines, and reagent logistics aligned to daily test volume.
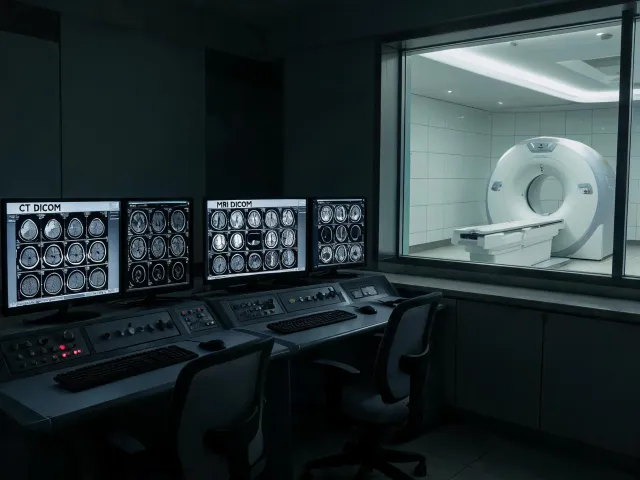
Modality selection with PACS, RIS, dose governance, and preventive maintenance requirements visible early.

UDI records, service history, cybersecurity patch cadence, and loaner coverage presented in one support plan.
Why teams ask Danaher first
Regulatory packets include 510(k) references when applicable, CE MDR declarations, IFU access, warranty terms, and UDI guidance for value analysis review.
DICOM, HL7 v2, FHIR R4, LIS, PACS, and EHR questions are captured before quoting so IT and clinical leaders do not inherit surprise work.
Short role-based modules help operators, biomed technicians, infection prevention teams, and department leads learn exactly what they need.
Urban, rural, and multi-site programs receive different response assumptions, spare-part stocking models, and escalation pathways.
Anonymous buyer notes
"The useful part was not a glossy demo. It was seeing service, IFU, and interface questions answered in one review."
"Our lab needed a plain discussion about throughput, QC, reagent stability, and LIS handoff. Danaher kept that conversation grounded."
"Dental equipment decisions move faster when training, sterilization notes, and multi-site support are visible before capital approval."
Buying questions
Yes. Danaher prepares separate clinical-use, technical-integration, and purchasing summaries so each stakeholder can review the evidence most relevant to their decision.
The team documents site location, device criticality, loaner needs, spare-part availability, and escalation contacts before finalizing a service recommendation.
Most teams ask for IFU access, regulatory status, warranty terms, cybersecurity statements, interoperability notes, and preventive maintenance schedules.
Yes. Use the Care Setting Finder to compare how dental, lab, and imaging systems fit clinics, hospitals, outpatient centers, and distributed networks.
